Our Pipeline:
A precision therapy.
A purpose-built mission.
Our Pipeline:
A precision therapy. A purpose-built mission.
Engineering Innovation for Pediatric Eyes
At Sydnexis, we’re advancing science with purpose. Our lead investigational product, SYD-101, is being developed as the first pharmaceutical treatment designed to help slow the progression of pediatric myopia. Engineered specifically for growing eyes, SYD-101 is a low dose, atropine eye drop. Administered once nightly, SYD-101 is being studied for use in children as young as 3 years old.
Unlike compounded atropine, which is widely used but not FDA-approved, SYD-101 aims to address known limitations such as formulation instability, inconsistent concentration, and variable bioavailability. While compounded medications are used today in the U.S, these limitations can introduce safety and efficacy risks that are less tolerable in the pediatric population.

SYD-101 is anticipated to be the first and only FDA-approved pharmaceutical treatment for pediatric progressive myopia for children ages 3-14.
The STAR Study: A Landmark Study Supporting the Future of Pediatric Progressive Myopia
Our STAR study is the largest Phase 3 clinical trial conducted to date in pediatric myopia management, enrolling 852 children across 47 sites. The study was designed to evaluate product safety and efficacy over three years, followed by a fourth-year randomized withdrawal phase.

The STAR Study: A Landmark Study Supporting the Future of Pediatric Progressive Myopia
Our STAR study is the largest Phase 3 clinical trial conducted to date in pediatric myopia management, enrolling 852 children across 47 sites. The study was designed to evaluate product safety and efficacy over three years, followed by a fourth-year randomized withdrawal phase.
Study Highlights:
The STAR study establishes a robust clinical foundation for SYD-101 and sets a new benchmark for evidence-based pharmaceutical innovation in myopia care.

June 30, 2024


Statistically significant reductions in myopia progression at 12, 24, and 36 months


Children aged 3–14 at treatment initiation


Year 4

3-year pivotal trial completed:
June 30, 2024

Primary endpoints met:
Statistically significant reductions in myopia progression at 12, 24, and 36 months

Patient population:
Children aged 3–14 at treatment initiation

Ongoing randomized withdrawal:
Year 4
Addressing an Unmet Need – Especially in Children Younger Than 10
Today’s treatment landscape for pediatric progressive myopia leaves a significant gap — particularly for younger children. Glasses correct refractive error but do nothing to address the underlying disease progression. While certain FDA-approved specialty contact lenses have demonstrated efficacy in controlling progression, they are not indicated for younger children.
SYD-101 is being developed to fill this gap as the first and only pharmaceutical treatment designed specifically to slow the progression of myopia in children as young as 3 years old. SYD-101 has the potential to bring a new standard of care to an underserved population — delivering long-term disease modification in a form that fits into real-world pediatric care.
SYD-101 Development Status
SYD-101 has progressed through all phases of clinical development and is now under regulatory review in the United States, with full approval already granted in EMEA. This timeline reflects Sydnexis’ commitment to rigorous science, regulatory excellence, and global impact.
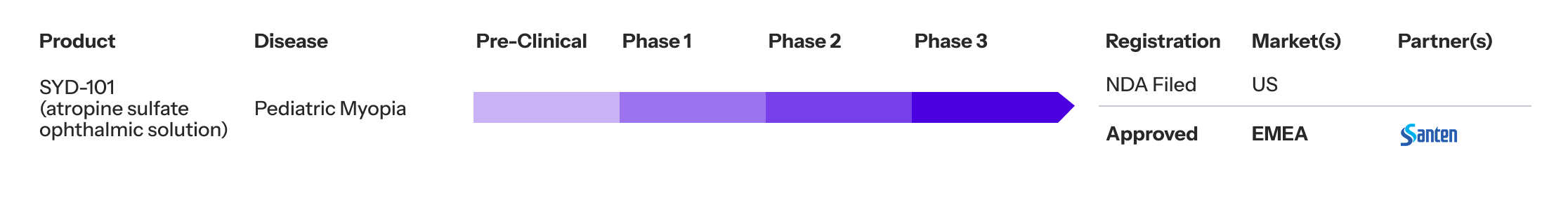
If approved, SYD-101 would be the only FDA-approved pharmaceutical treatment for pediatric progressive myopia for children as young as 3.
Be Part of a New Era in Myopia Treatment
"*" indicates required fields
